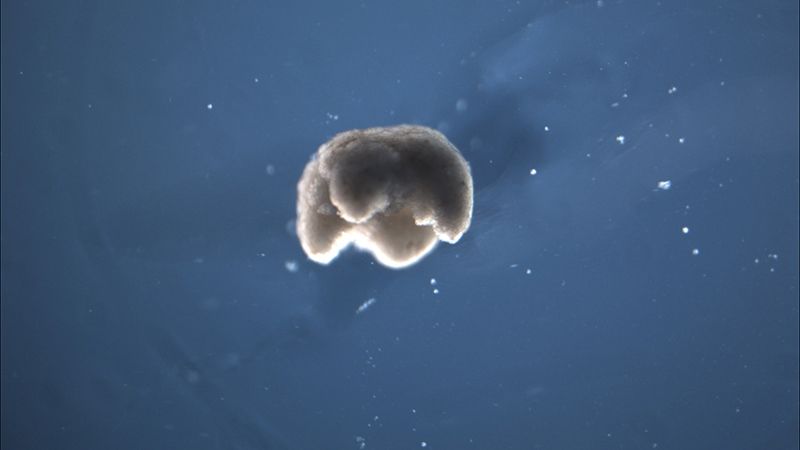
A manufactured quadruped measuring 650 to 750 microns in diameter — just a little smaller than the head of a pin — was built from amphibian stem cells.
(Image: © Douglas Blackiston, Tufts University)
What happens when you take cells from frog embryos and grow them into new organisms that were "evolved" by algorithms? You get something that researchers are calling the world's first "living machine."Though the original stem cells came from frogs — the African clawed frog, Xenopus laevis — these so-called xenobots don't resemble any known amphibians. The tiny blobs measure only 0.04 inches (1 millimeter) wide and are made of living tissue that biologists assembled into bodies designed by computer models, according to a new study.
These mobile organisms can move independently and collectively, can self-heal wounds and survive for weeks at a time, and could potentially be used to transport medicines inside a patient's body, scientists recently reported.
"They're neither a traditional robot nor a known species of animal," study co-author Joshua Bongard, a computer scientist and robotics expert at the University of Vermont, said in a statement. "It's a new class of artifact: a living, programmable organism."
Algorithms shaped the evolution of the xenobots. They grew from skin and heart stem cells into tissue clumps of several hundred cells that moved in pulses generated by heart muscle tissue, said lead study author Sam Kriegman, a doctoral candidate studying evolutionary robotics in the University of Vermont's Department of Computer Science, in Burlington.
"There's no external control from a remote control or bioelectricity. This is an autonomous agent — it's almost like a wind-up toy," Kriegman told Live Science.
Biologists fed a computer constraints for the autonomous xenobots, such as the maximum muscle power of their tissues, and how they might move through a watery environment. Then, the algorithm produced generations of the tiny organisms. The best-performing bots would "reproduce" inside the algorithm. And just as evolution works in the natural world, the least successful forms would be deleted by the computer program.
"Eventually, it was able to give us designs that actually were transferable to real cells. That was a breakthrough," Kriegman said.
The study authors then brought these designs to life, piecing stem cells together to form self-powered 3D shapes designed by the evolution algorithm. Skin cells held the xenobots together, and the beating of heart tissue in specific parts of their "bodies" propelled the 'bots through water in a petri dish for days, and even weeks at a stretch, without needing additional nutrients, according to the study. The 'bots were even able to repair significant damage, said Kriegman.
"We cut the living robot almost in half, and its cells automatically zippered its body back up," he said.
"We can imagine many useful applications of these living robots that other machines can't do," said study co-author Michael Levin, director of the Center for Regenerative and Developmental Biology at Tufts University in Massachusetts. These might include targeting toxic spills or radioactive contamination, collecting marine microplastics or even excavating plaque from human arteries, Levin said in a statement.
Creations that blur the line between robots and living organisms are popular subjects in science fiction; think of the killer machines in the "Terminator" movies or the replicants from the world of "Blade Runner." The prospect of so-called living robots — and using technology to create living organisms — understandably raises concerns for some, said Levin.
"That fear is not unreasonable," Levin said. "When we start to mess around with complex systems that we don't understand, we're going to get unintended consequences."
Nevertheless, building on simple organic forms like the xenobots could also lead to beneficial discoveries, he added.
"If humanity is going to survive into the future, we need to better understand how complex properties, somehow, emerge from simple rules," Levin said.
The findings were published online Jan. 13 in the journal Proceedings of the National Academy of Sciences.
Originally published on Live Science.
No comments:
Post a Comment